Atrophic Rhinitis
Definition:
Atrophic rhinitis is defined as a chronic nasal disease characterised by progressive atrophy of the nasal mucosa along with the underlying bones of turbinates. There is also associated presence of viscid secretion which rapidly dries up forming foul smelling crusts. This fetid odor is also known as ozaena. The nasal cavity is also abnormally patent. The patient is fortunately unaware of the stench emitting from the nose as this disorder is associated with merciful anosmia.
Aetiology:
The etiology of this problem still remains obscure. Numerous pathogens have been associated with this condition, the most important of them are
1. Coccobacillus,
2. Bacillus mucosus
3. Coccobacillus foetidus ozaenae
4. Diptheroid bacilli
5. Klebsiella ozaenae.
These organisms despite being isolated from the nose of diseased patients have not categorically been proved as the cause for the same.
Other possible factors which could predispose to this disease are:
1. Chronic sinusitis
2. Excessive surgical destruction of the nasal mucosa and turbiantes
3. Nutritional deficiencies
4. Syphilis.
5. Endocrine imbalances (Disease is known to worsen with pregnancy / menstruation)
6. Heredity (Autosomal dominent pattern of inheritance identified)
7. Autoimmune disease
The triad of atrophic rhinitis as described by Dr. Bernhard Fraenkel are 1. Fetor, 2. crusting and
3. atrophy.
Age of onset: Usually commences at puberty.
Females are commonly affected than males. Heredity is known to be an important factor as there appears to be increased susceptibility among yellow races, latin races and American negro races. Poor nurtrition could also be a factor. Bernat (1965) postulated iron deficiency could be a cause of this disorder.
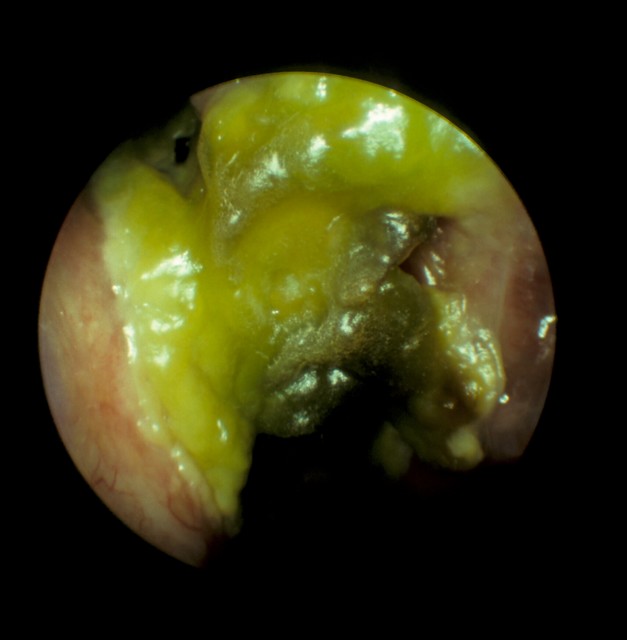
Greenish yellow crusts seen inside the nasal cavity of a patient with atrophic rhinitis
Recently immunologists have considered atrophic rhinitis to be an autoimmune disorder. Fouad confirmed that there was altered cellular reactivity, loss of tolerance to nasal tissues. This according to him could be caused / precipitated by virus infection, malnutrition, immunodeficiency.
Pathology:
1. Metaplasia of ciliated columnar nasal epithelium into squamous epithelium.
2. There is a decrease in the number and size of compound alveolar glands
3. Dilated capillaries are also seen
Pathologically atrophic rhinitis has been divided into two types:
Type I: is characterised by the presence of endarteritis and periarteritis of the terminal arterioles. This could be caused by chronic infections. These patients benefit from the vasodilator effects of oestrogen therapy.
Type II: is characterised by vasodilatation of the capillaries, these patients may worsen with estrogen therapy. The endothelial cells lining the dilated capillaries have been demonstrated to contain more cytoplasm than those of normal capillaries and they also showed a positive reaction for alkaline phosphatase suggesting the presence of active bone resorption. It has also been demonstrated that a majority of patients with atrophic rhinitis belong to type I category.
Once the diagnosis of atrophic rhinitis is made then the etiology should be sought. Atrophic rhinitis can be divided in to two types clinically:
1. Primary atrophic rhinitis - the classic form which is supposed to arise denovo. This diagnosis is made by a process of exclusion. This type of disease is still common in middle east and India. All the known causes of atrophic rhinitis must be excluded before coming to this diagnosis. Causative organisms in these patients have always be Klebsiella ozenae.
2. Secondary atrophic rhinitis: Is the most common form seen in developed countries. The most common causes for this problem could be:
1. Extensive destruction of nasal mucosa and turbinates during nasal surgery
2. Following irradiation
3. Granulomatous infections like leprosy, syphilis, tuberculosis etc
Clinical features:
The presenting symptoms are commonly nasal obstruction and epistaxis. Anosmia i.e. merciful may be present making the patient unaware of the smell emanating from the nose. These patients may also have pharyngitis sicca. Choking attacks may also be seen due to slippage of detached crusts from the nasopharynx into the oropharynx. These patients also appear to be dejected and depressed psychologically.
Clinical examination of these patients show that their nasal cavities filled with foul smelling greenish, yellow or black crusts, the nasal cavity appear to be enormously roomy. When these crusts are removed bleeding starts to occur.
Why nasal obstruction even in the presence of roomy nasal cavity?
This interesting question must be answered. The nasal cavity is filled with sensory nerve endings close to the nasal valve area. These receptors sense the flow of air through this area thus giving a sense of freeness in the nasal cavity. These nerve endings are destroyed in patients with atrophic rhinitis thus depriving the patient of this sensation. In the absence of these sensation the nose feels blocked.
Radiographic findings:
Are more or less the same in both primary and secondary atrophic rhinitis. Plain xrays show lateral bowing of nasal walls, thin or absent turbinates and hypoplastic maxillary sinuses.
CT scan findings:
1. Mucoperiosteal thickening of paranasal sinuses
2. Loss of definition of osteomeatal complex due to resorption of ethmoidal bulla and uncinate process
3. Hypoplastic maxillary sinuses
4. Enlargement of nasal cavity with erosion of the lateral nasal wall
5. Atrophy of inferior and middle turbinates
Management:
Conservative:
Nasal douching - The patient must be asked to douche the nose atleast twice a day with a solution prepared with:
Sodium bicarbonate - 28.4 g
Sodium diborate - 28.4 g
Sodium chloride - 56.7 g
mixed in 280 ml of luke warm water.
The crusts may be removed by forceps or suction. 25% glucose in glycerin drops can be applied to the nose thus inhibiting the growth of proteolytic organism.
In patients with histological type I atrophic rhinitis oestradiol in arachis oil 10,000 units/ml can be used as nasal drops.
Kemecetine antiozaena solution - is prepared with chloramphenicol 90mg, oestradiol dipropionate 0.64mg, vitamin D2 900 IU and propylene glycol in 1 ml of saline.
Potassium iodide can be prescribed orally to the patient in an attempt to increase the nasal secretion.
Systemic use of placental extracts have been attempted with varying degrees of success.
Surgical management:
1. Submucous injections of paraffin, and operations aimed at displacing the lateral nasal wall medially. This surgical procedure is known as Lautenslauger's operation.
2. Recently teflon strips, and autogenous cartilages have been inserted along the floor and lateral nasal wall after elevation of flaps.
3. Wilson's operation - Submucosal injection of 50% Teflon in glycerin paste.
4. Repeated stellate ganglion blocks have also been employed with some success
5. Young's operation - This surgery aims at closure of one or both nasal cavities by plastic surgery. Young's method is to raise folds of skin inside the nostril and suturing these folds together thus closing the nasal cavities. After a period of 6 to 9 months when these flaps are opened up the mucosa of the nasal cavities have found to be healed. This can be verified by postnasal examination before revision surgery is performed. Modifications of this procedure has been suggested (modified Young's operation) where a 3mm hole is left while closing the flaps in the nasal vestibule. This enables the patient to breath through the nasal cavities. It is better if these surgical procedures are done in a staged manner, while waiting for one nose to heal before attempting on the other side.
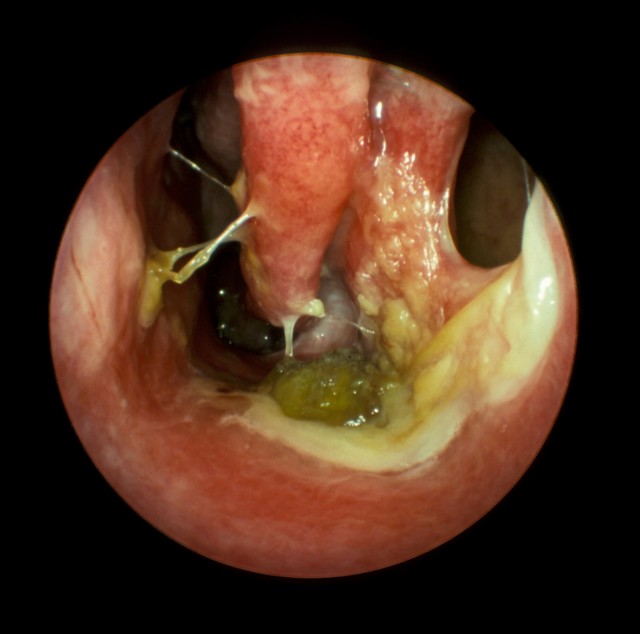
Endoscopic view of post operative atrophic rhinitis
